Real time Clinical Trial Data Visualization
Transform Data into Insight - As it happens
CPS Data Visualization provides real‑time, interactive insight into early‑phase study data, enabling faster, more confident decision‑making through live connectivity with ClinSpark®
Elevating Clinical Trials with Real-Time Data Visualization
By leveraging the combined eSource and EDC platform powered by ClinSpark®, Fortrea’s real‑time data visualization delivers live access to study data as it is being collected. Data Visualization is a powerful add‑on to Fortrea’s Clinical Pharmacology Services, enabling sponsors to monitor their data, identify trends or anomalies, and make fast and informed decisions. This is achieved without the need to wait for end-of-study reports, bringing valuable insights to all stakeholders promptly upon data collection.
A Centralized, Interactive Dashboard
All visualisations are bespoke, customisable, and interactive, and housed within a centralised clinical data dashboard for all authorised stakeholders to access.
Real-Time Collaboration
A shared real‑time dashboard supports better collaboration, maintains data quality, and enables early sponsor decision‑making.
Know-how at the Core
Ahead of study start-up, Fortrea collaborate with SMEs to design dashboards optimally aligned to protocol-specific endpoints.
The dashboard is available to sponsors and tailored to their specific requirements
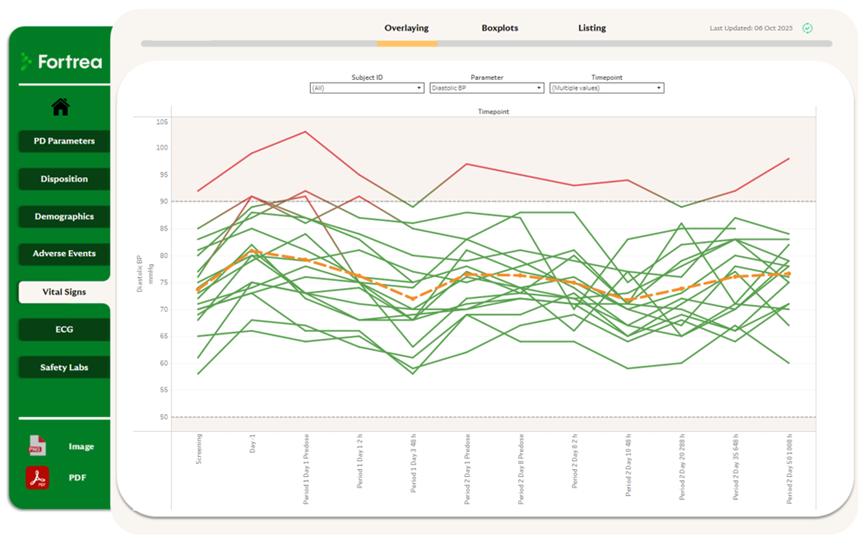
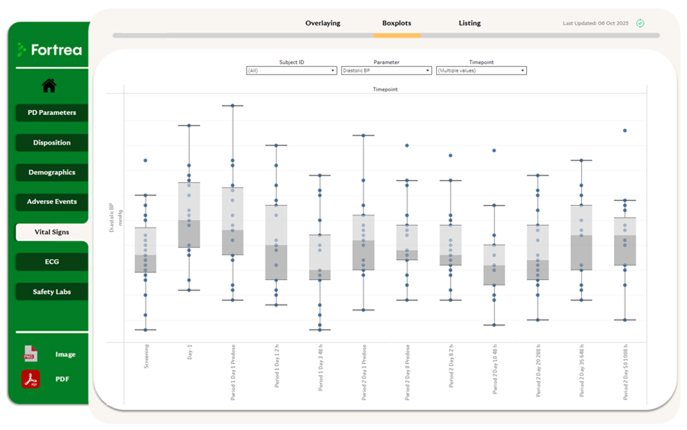
Why Near Real‑Time Matters in Clinical Trials?
In today’s fast-moving development environment, delayed visibility can slow progress and obscure risk.
Fortrea’s approach to real-time visualization provides:
Direct source connectivity with 10-minute data refreshes from ClinSpark® – enabling earlier insight into trends and risk.
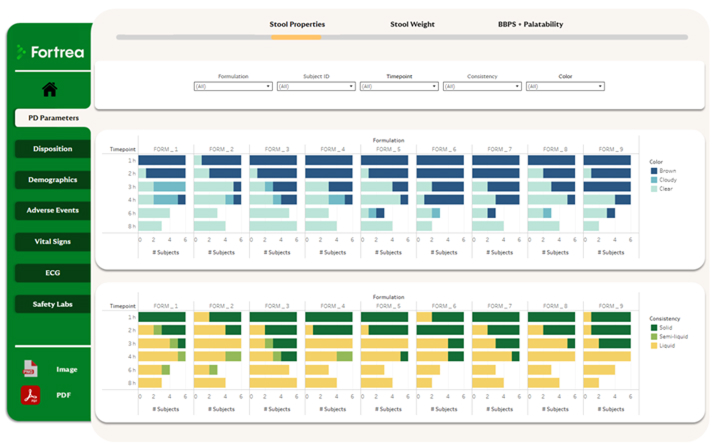
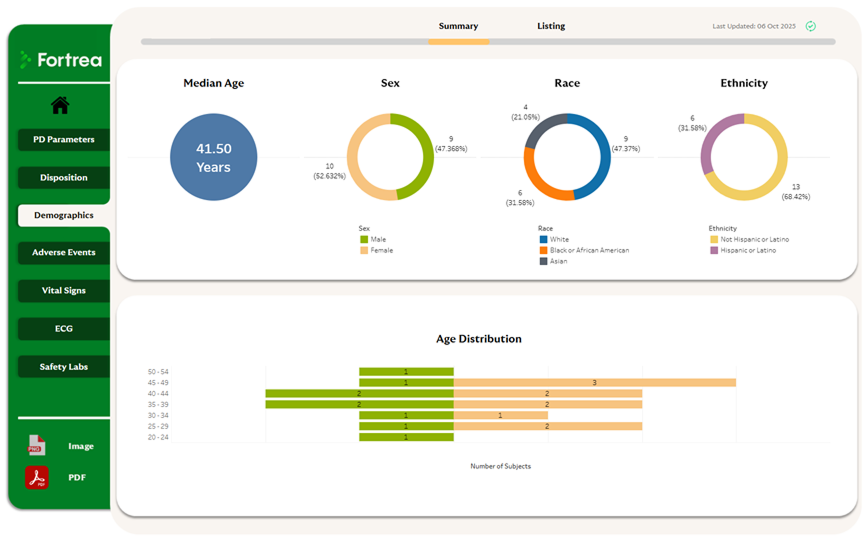
Three Levels of Data Insight
Fortrea delivers data visibility at multiple levels to support both operational and strategic oversight:
Participant level – Individual participant profiles track study progression and biometric data
Project level – Aggregated study views aligned to key endpoints and milestones
Portfolio level – Consolidated oversight across studies, investigational products, therapeutic areas, or sites
Connectivity for Continuous Data Updates
At Fortrea CRUs, the Fortrea Data Visualization Engine connects directly to the ClinSpark® EDC database, refreshing every 10 minutes to provide a near real‑time view of ongoing data collection.
Ancillary data sources are integrated into the same dashboard within 24 hours of data transfer receipt.
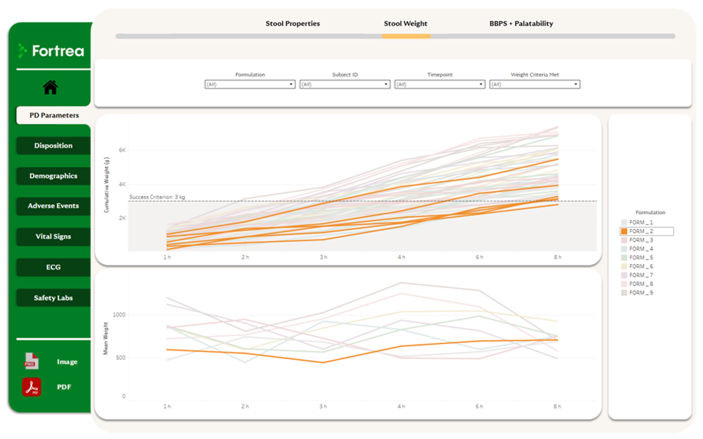
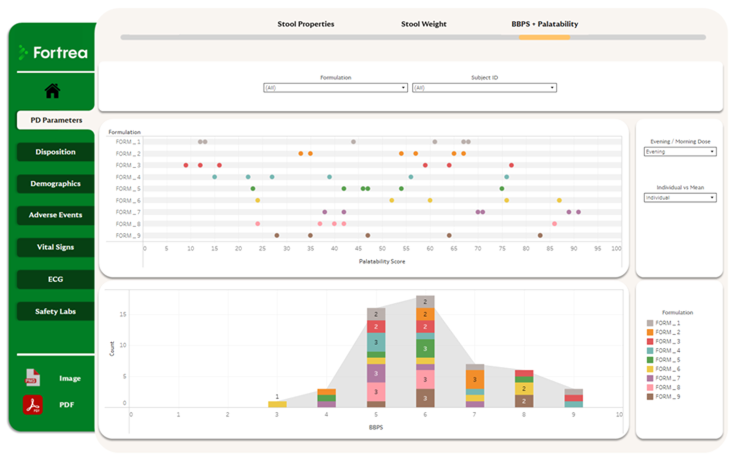
A Unified Clinical Data Experience
By bringing multiple data sources together in a single environment, users can quickly navigate between data points without switching tools - streamlining oversight and analysis
| Clarity at a Glance | Faster Detection of Risk and Opportunity | Flexible Access and Export |
|---|---|---|
| Clear, high‑level visualizations make study status easy to understand and act upon. | Interactive graphics reveal trends and anomalies earlier for faster decisions | Filtered, exportable insights are ready for presentations and key decisions |

